Introduction
Currently, over two million people around the world suffer from end-stage renal disease (ESRD). The best treatment for current patients with ESRD is a kidney transplant [
1]. This method of treatment is severely limited by donor organ availability. In 2018 alone, over 100,000 people in the United States were on the kidney transplant waiting list, while only 21,000 organs were available for transplant [
1]. The need for donor organs in the United States is predicted to rise by 8% each year [
2]. The current standards of care for patients with kidney failure or ESRD include full kidney transplant with a donor organ, in-center hemodialysis (HD) or peritoneal dialysis (PD), or at-home HD via an external machine. The severe lack of donor kidneys leads to extended dialysis treatments as the norm to treat patients with ESRD. Long-term dialysis is associated with several comorbidities, including an increased risk for kidney cancer. Additionally, patients who receive a kidney transplant have been found to be at higher risk for cancer in general due to the immunosuppressant drugs required after transplant surgery [
3].
The field of kidney replacement technology has evolved greatly over the last two decades, with improvements in nanotechnology, cell growth techniques, and bioreactors. Two of the most recent technological advancements in this field are the implantable bioartificial kidney (BAK) and kidney regeneration technology. Both techniques are in preclinical stages and aim to fully replace normal kidney functionality. Both technologies address donor organ shortages as well as complications from dialysis and immunosuppressants. The purpose of this review is to analyze recent progress in kidney replacement technology and assess its potential impact on reducing risks associated with dialysis and donor organ kidney transplants, specifically donor shortages, renal failure, and risk of cancer.
History of renal replacement technology
Historically, kidney failure following ESRD is best treated by a full kidney transplant with a donor organ. Given the extremely limited availability of donor organs, most patients with a failing kidney end up on dialysis, either HD or PD. The problem with dialysis is that it is suboptimal in terms of morbidity and mortality. While dialysis accounts for kidney filtration function of small solute clearance, it does not make up for the loss of metabolic, endocrine, and reclamation functions of the kidney, resulting in poor outcomes. Innovations in renal replacement technology have been a growing focus over the last two decades, aiming to create a product that will replace full kidney functionality, not just the filtration aspect. Two less recent models of renal replacement technology are the automated wearable artificial kidney (AWAK) and the wearable artificial kidney (WAK) (
Fig. 1ŌĆō
3) [
4].
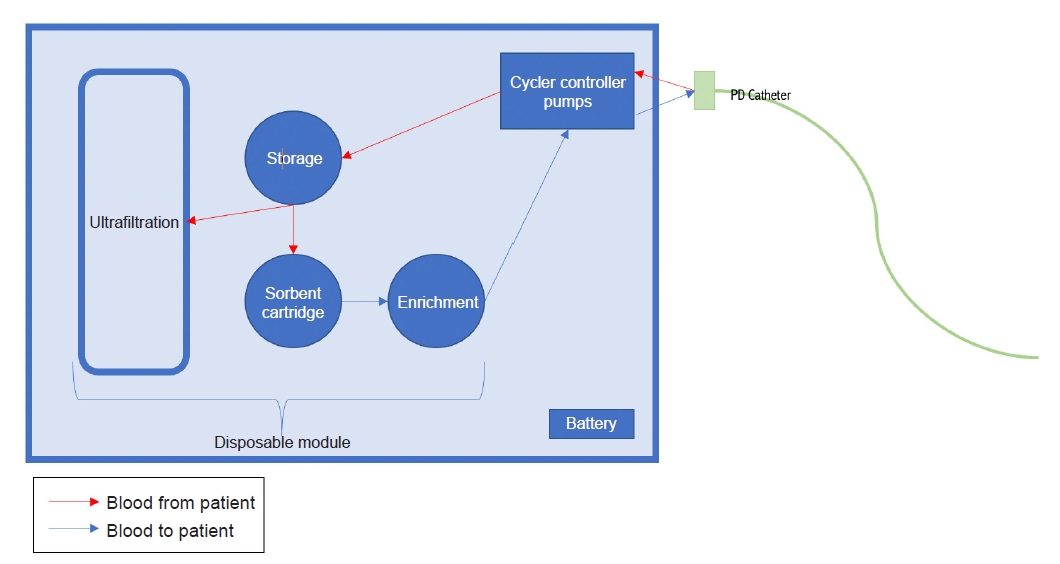
The AWAK is a tidal PD-based artificial kidney that uses dialysate regeneration to reduce fluid requirements. It consists of tubing, a disposable storage module, and a system controller compacted into a device the size of an average handbag. Dialysate (the reserve volume) is instilled into the peritoneal cavity and absorbs toxins, waste products, and fluid through the peritoneal membrane. The reserve volume of the regenerated dialysate is returned to the peritoneal cavity, and the remaining fluid (ultrafiltrate) is drained into an ultrafiltration bag and can be discarded. The procedure can be repeated after replacing the used cartridge with a new one. This device has been approved for trials in humans [
4].
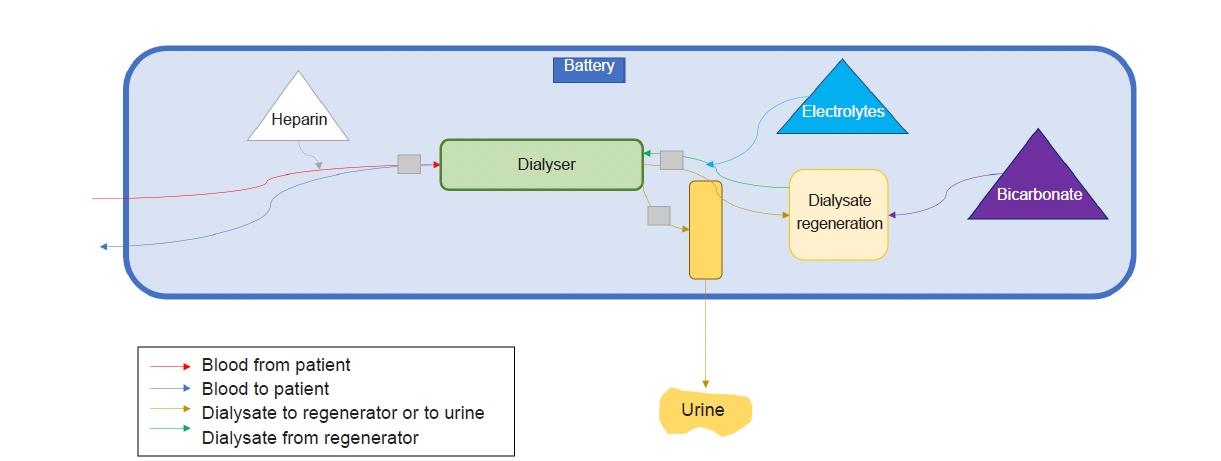
The WAK is a portable blood-based renal replacement device that is battery operated and can be worn like a belt or vest. The blood flowing through this system is anticoagulated with heparin using a syringe pump and then moves on to a two-channel pump that alternately propels blood and dialysate to a small dialyzer. Next, blood exits the dialyzer and travels through a bubble detector before being returned to the patient. The dialysate is regenerated for further use. An ultrafiltration pump controls fluid removal by portioning off a part of the regenerated dialysate to a waste bag for removal. This device is U.S. Food and Drug Administration (FDA)-approved for clinical trials [
4].
The major limitation in both current devices is that they still do achieve full kidney functionality; they are focused on ultrafiltration and remain lifestyle limiting, albeit less so than traditional dialysis. They require external machinery and systems, which limit mobility for the patient using it. Additionally, the lack of endocrine and metabolic functionality causes poorer outcomes for dialysis patients over time compared with patients who received a full transplant.
The two newest iterations in renal replacement technology, the implantable BAK and kidney regeneration technology, address the limitations of the AWAK and WAK devices in that both aim to provide full kidney functionality as well as improved patient mobility and autonomy.
Implantable bioartificial kidney
State of technology
The implantable BAK will provide another alternative for ESRD patients. This device not only reduces time on dialysis, but also replaces total kidney functionality. The filtration component of kidney function occurs at the glomerulus. Ultrafiltration of the blood is performed to remove toxic waste from circulation and retain important materials within systemic circulation, such as albumin. The regulatory component of the kidney occurs at tubular segments attached to the glomerulus. Ultrafiltrate from the glomerulus moves along the kidney tubule, which reabsorbs fluid and solutes to finely regulate the excretion of various amounts of solutes and water in urine. Both of these functionalities are necessary in a fully functional kidney unit. The implantable BAK combines a high efficiency filter connected in series with a bioreactor of cultured renal tubule epithelial cells to achieve classification as a fully functioning kidney unit (
Fig. 4,
5) [
5,
6].
The implantable BAK achieves solute transfer with convective transport, which is independent of concentration gradient and instead depends on a hydraulic pressure gradient across a membrane. This method of transport for toxin removal is advantageous because it mimics the natural glomerular process of toxin clearance of solutes with a higher molecular weight and solutes of the same diffusion rate. Convective transport in an implantable device can be achieved with polysulfone hollow fibers, which can be lined with renal endothelial cells and placed into the arteriovenous circuit using the common iliac artery and vein [
6]. This arteriovenous connection allows the device to operate on blood pressure rather than an externally- or battery-powered pump.
Designing the technology
The first step toward a fully implantable BAK is to perfect the combination of hemofilter and bioreactor devices in an extracorporeal setting. In 2002, a working prototype of the BAK was created with the development of an extracorporeal device that consisted of a hemofiltration cartridge containing over 109 renal tubule cells grown as monolayers along the inner surface of the fibers. These hollow fibers act as scaffolds for the renal tubule cells because they are non-biodegradable and have an optimal pore size for an immunoprotective barrier.
In vitro renal tubule assist device (RAD) studies showed that the cells retained differentiated active transport properties, differentiated metabolic activities, and important endocrine processes. The studies also showed that the device replaces endocrine, filtration, transport, and metabolic kidney functions when connected in series with a blood filtration device [
7].
In 2004, Phase I and Phase II clinical trials were approved by the FDA for assessing the response of 10 patients with acute renal failure and multiple organ failure to a BAK device. A synthetic hemofilter was connected in series with a bioreactor cartridge that contained around 109 human proximal tubule cells, acting as a RAD, within an extracorporeal perfusion circuit utilizing standard hemofiltration pump systems. The results showed 6 of 10 patients surviving past 30 days [
8].
The kidney was the first solid organ whose partial functionality was performed by a machine, with installment of HD and PD, as well as the first organ to be successfully transplanted as an isograft [
8]. As mentioned prior, the major limitations of HD and PD are that they do not provide regulatory, homeostatic, metabolic, and endocrine functions of the kidney, and their long-term use is associated with an increased risk of acquired cystic kidney disease (ACKD), various infections, and even cancer [
9]. A fully functioning kidney needs the filtering function of the glomerulus and the regulatory/transport capability of the tubule. Existing technology has addressed glomerular and excretory functions but lacks the regulatory and endocrine functionality. The initial focus in kidney tissue engineering was to develop an extracorporeal bioartificial device that would consist of a conventional synthetic hemofilter with a RAD. As of 2013, proof of concept for a wearable BAK was achieved by combining existing PD technology with a bioartificial renal epithelial system [
10]. The renal epithelial system is needed because tubular functionality of the kidneys cannot be replaced with inanimate devices as can filtration functionality. Instead, tubular functionality requires the naturally evolved biological membrane of the renal tubular epithelium. A bioartificial tubule was constructed with renal tubule progenitor cells seeded onto semipermeable hollow-fiber membranes that have been layered with an extracellular matrix (ECM) to enhance the attachment and growth of epithelial cells. These membranes provided both a scaffold for the cells to grow on and immunoprotection, similar to what has been documented for bioartificial pancreas implantation in a xenogenic host [
10,
11]. The next step in the process was to improve the wearable bioartificial technology to be implantable and more fully mimic a donor kidney. While the wearable device improves quality life in terms of mobility and autonomy as well as returning full kidney functionality to the patient, it falls short of an implantable device that can exist autonomously inside the body. The implantable device consists of a silicon hemofilter for toxin filtration and a bioreactor of renal epithelial cells for metabolic and endocrine functions. The filters in natural kidneys are elongated, ŌĆśslit-shapedŌĆÖ structures that optimize the permeability and selectivity tradeoff. In the implantable BAK, silicon nanotechnology is used in the hemofilter structure to imitate the membrane of a natural kidney.
Limitations and challenges
A major limitation in creating the implantable BAK is the miniaturization aspect. An avenue that has been explored to create the device prototype is microelectromechanical systems (MEMS) (
Fig. 6). MEMS is an industrial toolkit that applies mature manufacturing techniques from the semiconductor industry to miniature electromechanical devices, such as pumps, valves, and sensors. This technology can be used to produce silicon membranes containing ŌĆśslit-shapedŌĆÖ pores that are necessary for producing an implantable BAK [
10]. Another engineering challenge for the implantable BAK is to design it such that the membrane maximizes water permeability while minimizing leakage of albumin and other important macromolecules. This challenge is overcome using silicon nanotechnology slit pores. A challenge in using silicon is the oxide coating that can form when exposed to oxygen. The coating can be prevented by modifying the silicone surface with a highly hydrated polymer by grafting an organic polymer to the silicon nanopore surface [
10,
11]. A long-term challenge for the implantable device is combating coagulation; a sustainable anticoagulation solution will be essential for a fully implantable BAK device. Further limitations of the implantable device are the size and pump requirements of modern dialyzers, and the water volume required for dialytic therapy [
11].
Novel renal replacement technological components: BioCartridge and HemoCartridge
A newly pioneered collaboration among multiple academic institutions is working on creating the first fully functioning, FDA-approved implantable BAK. The device contains both a ŌĆ£HemoCartridgeŌĆØ and ŌĆ£BioCartridge,ŌĆØ where the HemoCartridge is a high efficiency filter that utilizes the silicon slit-pore nanotechnology and the BioCartridge is a bioreactor containing cultured renal tubule epithelial cells. Ultrafiltrate is generated in the HemoCartridge component and then flows to the BioCartridge component where it is processed to return salt, water, and glucose to the blood and filter toxins into a small volume of fluid comparable to urine. The implantable BAK is connected directly to the patientŌĆÖs vasculature and does not require an electrical pump due to connection to the circulatory system. Blood pressure pumps the blood through the device starting at the HemoCartridge via the membranes that imitate the slit-shaped pores of podocytes. Then, the blood flows through the BioCartridge that contains living tubular cells to imitate the glomerulus functionality of the kidney [
5].
The implantable BAK device removes the need for dialysate since the reabsorption of salt and water in the BioCartridge maintains approximately neutral fluid balance with removal of concentrated waste. The silicon membranes in this device are coated with hydrated biocompatible polymers that protect blood from stagnation and excess shear, allowing anticoagulant-free clinical implantation [
5]. After achieving proof of concept with the HemoCartridge and BioCartridge device, this project is currently in the preclinical testing stage. A pilot study is being conducted to test the two components together once they are miniaturized. The next phase of development is clinical trials with human subjects.
Successful implementation of this device into clinical practice could drastically reduce the length of time a potential kidney transplant patient spends on dialysis due to the ability of the implantable BAK to supplement the small number of donor organs available each year. Additionally, since the BioCartridge component of the implantable BAK contains the patientŌĆÖs own cell line, it is more biologically compatible. Ideally, this device would lessen the need for the long-term immunosuppression that typically follows transplantation procedures.
Kidney regeneration technology
Another recent development in bioengineering kidney technology is cellular regeneration. Progress in stem cell and developmental biology has realized the vision of creating a transplantable kidney graft composed of a patientŌĆÖs own cells. Directed differentiation allows control of stem cell development through key milestones to create the building blocks for autologous kidney regeneration. Natural kidney development and regeneration need to be continuously studied to further understand the tissue regeneration process [
12].
The main method currently used in kidney generation involves scaffolding [
12,
13]. An ECM scaffold is used for three-dimensional structural support for vasculature and specific cell types of the organ. Creating scaffolds similar in complexity, structure, and size to human organs is a persistent challenge. Detergent-based perfusion decellularization of mammalian organs, which utilizes a carefully formulated solution to distill an organ down to the ECM scaffold, has been successfully applied to multiple organ systems, including the kidney [
12,
13]. However, kidney scaffolds of human origin require more thorough examination of composition, growth factor levels, and mechanical properties of native developing and adult kidneys. Further research will provide a better understanding of the components preserved versus lost during the decellularization process, and the results will provide a roadmap for building kidney scaffolds with optimal accommodation factors for the cell types necessary for organ functionality (
Fig. 7) [
13].
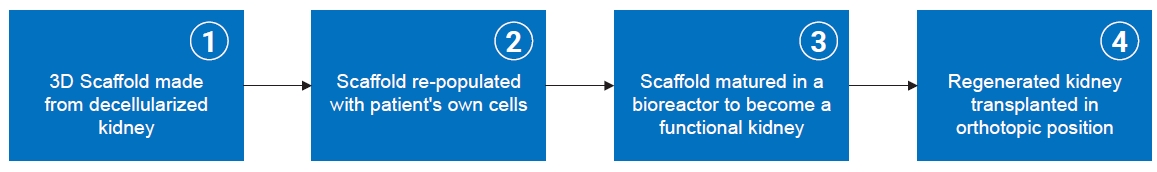
Success in decellularizing rat kidneys was achieved with detergent perfusion to create whole organ scaffolds with perfusable vascular, glomerular, and tubular compartments that can facilitate whole organ functionality. Rat kidneys decellularized using the perfusion method were shown to preserve structure and composition of the ECM, which is essential for filtration, secretion, and reabsorption [
14]. The decellularized scaffolds were repopulated with endothelial and epithelial cells grown from the ratŌĆÖs baseline stem cells to create functional kidney grafts. Then, the bioengineered kidneys were transplanted in orthotopic position, and urine production was found to be normal [
13,
14].
Kidney regeneration technology is currently in the preclinical trial stage. Overall, cadaveric kidneys can be decellularized to structural scaffolds, recellularized with endothelial and epithelial cells, matured in a bioreactor to a functional kidney, and transplanted in orthotopic position to provide normal kidney functions
in vivo. Progression of the technology will necessitate scaling of the cell seeding processes to larger human organ scaffolds. Perfusion decellularization techniques also have potential benefits in the three-dimensional (3D) bioprinting of kidney cells. In order to artificially 3D bioprint the cells necessary for a functional organ, a specific ŌĆ£bioinkŌĆØ is needed to create a particular setting that reinforces cellular growth and proliferation. A recent study demonstrated the viability of these decellularized ECM scaffolds from porcine kidneys to generate a hydrogel for the bioink of 3D-printed kidneys. This ECM-derived bioink improved cell growth and proliferation and even achieved organizational features of innate renal tissue [
15]. Successful implementation of this technology in clinical practice will likely eliminate the need for immunosuppressant prescriptions for kidney transplant patients since the kidneys are grown from the patientŌĆÖs own tissue. This technology will also likely decrease the amount of time ESRD patients remain on dialysis by supplementing the small pool of available donor organs each year.
Potential application to kidney cancer
As mentioned before, both implantable BAK and kidney regeneration technology have the potential to address donor organ shortages, the need for immunosuppression, and the shortcomings of previously designed renal replacement devices, such as mobility and full kidney functionality. Another issue they can address is the risk of cancer presentation, specifically renal cell carcinoma (RCC), in long-term dialysis patients and/or kidney transplant recipients.
In a population-based study of the United States' kidney transplants, over 100,000 kidney transplant recipients were observed. The risk of RCC in these kidney transplant recipients was 5.7-fold higher than that of the general population. The risk of papillary RCC was much higher than for clear cell RCC. The overall RCC risk was highest in recipients who had dealt with long-term dialysis before their transplant procedure. The vast majority (89%) of RCC occurrences in these cases occurred in the patientŌĆÖs native kidney [
9]. Additionally, a bimodal distribution of RCC onset after transplantation was observed, as a high risk for cancer was recorded in the time period immediately following transplant and another high-risk spike approximately 30 months after procedure. Following the second spike, the risk for RCC remains gradually increasing over time [
9].
It is hypothesized that some RCC tumors appear in the native kidney as a resulting complication of ACKD. The complication of ACKD that is associated with RCC development in the native kidney is renal cysts that transform into malignant cancers [
9]. According to the National Institute of Diabetes and Digestive and Kidney Diseases, 20% of patients who begin dialysis already have ACKD, between 60% and 80% of patients on dialysis for at least 4 years develop ACKD, and 90% of patients on dialysis for at least 8 years develop ACKD [
9]. Since most kidney transplant patients are on dialysis for extended periods of time before their transplant procedures due to the lack of donor organs, these patients subsequently have a higher probability of RCC associated with the renal cyst complication from ACKD.
Additionally, post-procedure transplant patients are put on a regimen of immunosuppressants, which are thought to be linked to higher susceptibility to cancer because these drugs decrease the ability of a patientŌĆÖs immune system to detect cancer cells or fight against infections that may cause cancer [
3,
9,
16]. Moreover, a study of 7,217 kidney transplant patients confirmed the hypothesis that kidney transplant recipients have higher risk of
de novo cancers, with non-Hodgkin lymphoma, lung, kidney, and prostate cancers as the most common types. That study associated the increased risk of cancer post-transplant with immunosuppressive drugs prescribed after the transplant procedure [
17].
Both previously mentioned kidney technologies have the potential to address the higher frequency of kidney cancer in transplant patients. The implantable BAK and kidney regeneration technique are theorized to reduce the need for immunosuppressants since these devices would be seeded from a patientŌĆÖs own tissue, lessening the overall immune response to the devices [
4,
18,
19]. Additionally, both technologies would inherently increase the amount of donor organs available, decreasing the time patients would spend on dialysis. An increase in transplantation accessibility in a timelier manner could potentially reduce the occurrence of ACKD in these patients since ACKD is associated with dialysis duration. A reduction in ACKD instances would subsequently reduce the instances of known ACKD-associated complications, one of which is kidney cancer (
Table 1).
Advancements in implantable BAK device and kidney regeneration technology could drastically improve the lives of hundreds of thousands of people on dialysis each year and save the lives of thousands who die each year waiting for a kidney. These technologies also have potential applications to the risk factor of kidney cancer that is associated with patients who receive a transplant.
Machine learning/artificial intelligence
The application of artificial intelligence (AI) and machine learning (ML) to medicine has gained traction in recent years. FDA has approved the use of AI in monitoring for atrial fibrillation, coronary calcium scoring, and diagnosis of CT brain bleeds [
20]. While renal regeneration and BAK are promising novel technologies, the advent of AI and ML has shown beneficial results with HD and kidney transplantation [
20]. Concepts such as artificial neural networks (ANN), internet of things, and ŌĆ£deepŌĆØ learning technology use extensive data that can predict and execute ŌĆ£personalizedŌĆØ medical decisions for each patient. These algorithms have been shown to better predict changes in HD, such as hypotension and heart rate variability/volumes, than nephrologists [
20]. These technologies can be programmed to interactively adapt and react to such complications that occur in HD in real time. This data-driven technology would be beneficial for patients to reduce additional unnecessary medications and the cost of any corrective interventions. Regarding kidney transplantation, ANN were able to better predict the probability of chronic renal allograft rejection, which can enable more precise allotment of organ transplants [
20].
The implementation of AI/ML in medicine is still years away as there is a paucity of studies proving its benefit in real-world patients. Additionally, issues with patient privacy and data security persist and must be dealt with before the use of such algorithms [
20]. As BAK and cellular kidney regeneration are at the forefront in kidney replacement technology, application of data-driven AI and ML concepts to former technologies such as dialysis or kidney transplantation may provide an alternative route for patients who are comfortable with remaining on their dialysis routine or are steadfast in waiting for a kidney transplant.
Conclusion
The two newest innovations in renal replacement technologyŌĆöimplantable BAK and cellular kidney regenerationŌĆöcreate a fully functioning alternative to long-term dialysis or a donor organ. They improve on previous iterations of renal replacement technology by accomplishing all aspects of normal kidney functionality, while also being fully implantable and autologous to allow patients maximum mobility. Additionally, these technologies may have the potential to address many associated risks of dialysis and kidney transplants, such as potential infections, effects of immunosuppression, and the risk of cancer - specifically renal cancer. As these technologies move out of preclinical testing stages into clinical testing and eventual clinical practice, they must be further studied to analyze their impact on instances of renal cancer in kidney transplant patients.












 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print















