A questionnaire survey on the diagnosis and treatment of Fabry nephropathy in clinical practice
Article information
Abstract
Background
Fabry nephropathy is characterized by a deficiency of lysosomal alpha-galactosidase A, which results in proteinuria and kidney disease. The ineffectiveness of enzyme replacement therapy (ERT) for severe kidney failure highlights the need for early detection and meaningful markers. However, because the diagnosis and treatment of Fabry disease can vary according to the expertise of physicians, we evaluated the opinions of Korean specialists.
Methods
A questionnaire regarding the management of Fabry nephropathy was emailed to healthcare providers with the experience or ability to treat individuals with Fabry nephropathy.
Results
Of the 70 experts who responded to the survey, 43 were nephrologists, and 64.3% of the respondents reported having treated patients with Fabry disease. Pediatricians are treating primarily patients with classic types of the disease, while nephrologists and cardiologists are treating more patients with variant types. Only 40.7% of non-nephrologists agreed that a kidney biopsy was required at the time of diagnosis, compared with 81.4% of nephrologists. Thirty-eight of 70 respondents (54.3%) reported measuring globotriaosylsphingosine (lyso-Gb3) as a biomarker. The most common period to measure lyso-Gb3 was at the time of diagnosis, followed by after ERT, before ERT, and at screening. For the stage at which ERT should begin, microalbuminuria and proteinuria were chosen by 51.8% and 28.6% of respondents, respectively.
Conclusion
Nephrologists are more likely to treat variant Fabry disease rather than classic cases, and they agree that ERT should be initiated early in Fabry nephropathy, using lyso-Gb3 as a biomarker.
Introduction
Fabry nephropathy is caused by a deficiency in lysosomal alpha-galactosidase A (α-Gal A), which leads to an accumulation of globotriaosylceramide (Gb3) in kidney cells and the increase of its metabolites including globotriaosylsphingosine (lyso-Gb3) [1]. Kidney involvement manifests as proteinuria and decreased glomerular filtration rate (GFR) values, both of which are indicative of chronic kidney disease [2,3]. Enzyme replacement therapy (ERT) ameliorates Gb3 deposition in kidneys [4,5]. However, ERT has been shown to be ineffective in patients with advanced chronic kidney disease (as indicated by proteinuria and a low GFR), emphasizing the need for early detection of Fabry nephropathy [6–9]. A change in Gb3 deposition in a kidney biopsy is a useful indicator of Fabry nephropathy, but not of the efficacy of treatment [10,11]. Plasma lyso-Gb3, which has also been proposed as a diagnostic biomarker, has been shown to correlate with disease severity, enzyme replacement response, and phenotyping [12,13].
Fabry disease is classified as either a classical or variant phenotype (later-onset) [14,15]. Classical phenotypes present in childhood or adolescence with typical symptoms, including angiokeratomas, anhidrosis, tinnitus, hearing loss, corneal dystrophy, strokes, left ventricular hypertrophy, cardiac arrhythmias, abdominal discomfort, and diarrhea. In contrast, variant phenotypes have residual α-Gal A activity and are not associated with early manifestations of classic symptoms. Patients with the variant phenotype experience an essentially normal childhood and adolescence, typically only developing renal or cardiac disease in the third to seventh decades of life [15].
The Korean medical insurance system and the Korea Disease Control and Prevention Agency maintain a rare-disease registry. Patients with Fabry disease are eligible for reimbursement of medical expenses if their diagnosis is confirmed. Despite these systems, individuals with variant phenotypic Fabry disease are commonly misdiagnosed and unable to receive treatment for an extended period of time due to a lack of typical symptoms; such patients are typically less seriously affected, and disease presentations may be limited to a single organ [16]. Half of the patients with Fabry disease in a report from Argentina were diagnosed by nephrologists [17].
Some patients with variant types who met the criteria for indication, which includes kidney manifestation, were covered by medical insurance for ERT. However, lyso-Gb3 was not covered by insurance in Korea [18].
The pattern of Fabry nephropathy management in Fabry disease has not yet been surveyed in Korea. This study was designed to determine how Korean experts treat patients with Fabry nephropathy.
Methods
This study was approved by and received a waiver for the need for informed consent from the Institutional Review Board of the Soonchunhyang University Bucheon Hospital (No. 2021-07-045-001).
A questionnaire to determine patterns in the diagnosis and treatment of Fabry nephropathy was distributed from 2021 to 2022. Questionnaires were emailed to and received by registered members of the Korean Society of Nephrology (KSN) and the Korean Society of Medical Genetics and Genomics (KSMG).
The questionnaire was divided into eight sections: 1) age group and sex of responder; 2) experience with Fabry disease, which includes yes/no questions in the past and present, and number and phenotype of patients; 3) timing of kidney biopsy (on diagnosis, before ERT, on a regular period, on time of proteinuria, or not necessary); 4) the need for a Fabry registry (yes vs. no); 5) check-up of lyso-Gb3 including the timing, interval, and needs for insurance coverage; 6) the interval and preferred tests for kidney function in Fabry disease; 7) the definition and tests of Fabry nephropathy; and 8) treatments other than ERT for Fabry nephropathy. Some detailed questions had multiple answers. The questionnaire is supplied in Supplementary Table 1 (available online).
Descriptive analytical statistics were used to summarize survey responses. The chi-square tests were used to compare categorical variables between groups. All statistical analyses were performed using the IBM SPSS Statistics version 20.0 software program (IBM Corp.). A p-value of less than 0.05 was considered statistically significant.
Results
We sent emails to all 1,990 registered members of the KSN and KSMG, and 70 responded. The highest participation rate was among those in their 40s (55.7%), and 32 participants (45.7%) were male. The majority were nephrologists (61.4%), followed by pediatricians and cardiologists. Table 1 summarizes the baseline characteristics of the participants.
Experience treating Fabry disease patients
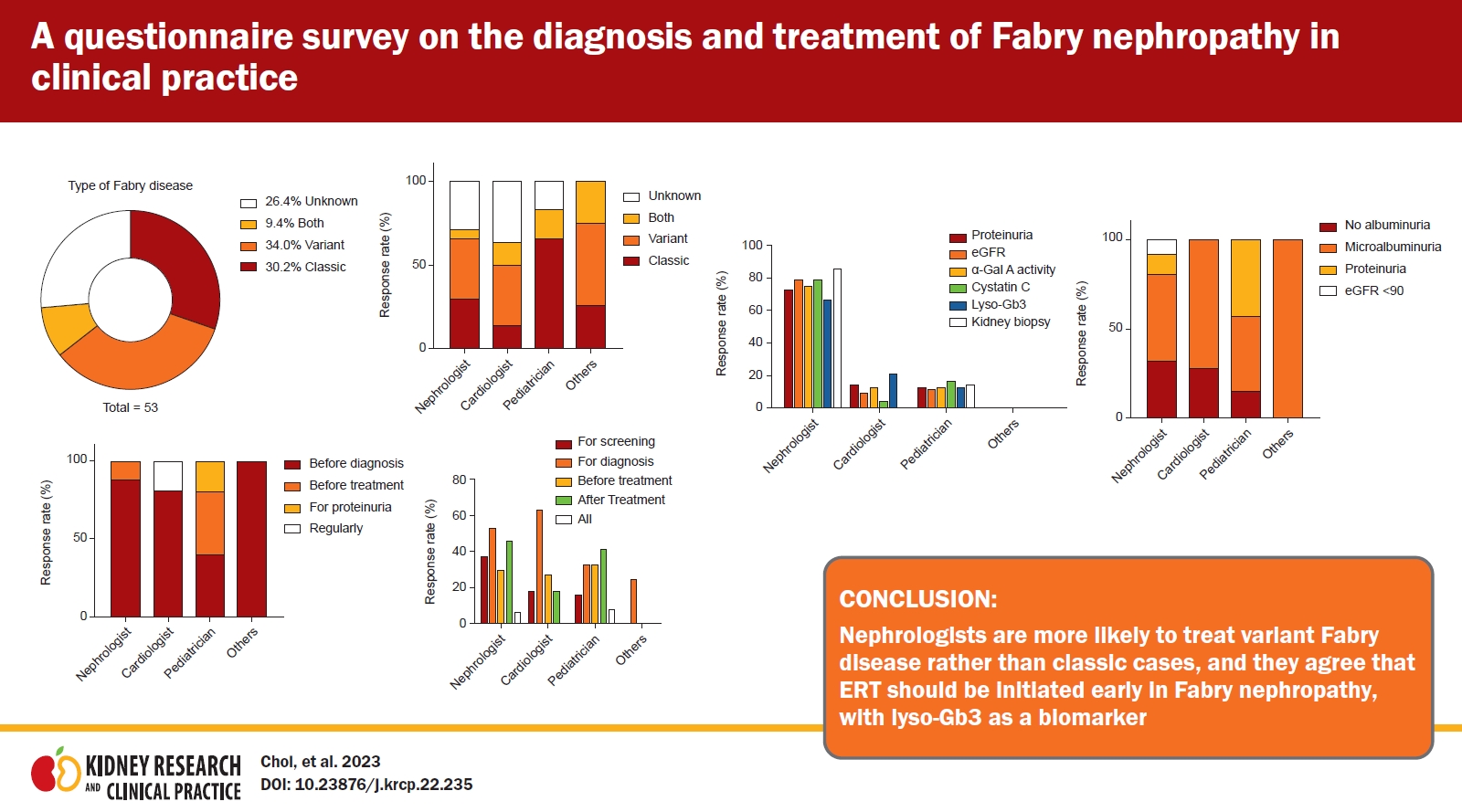
Sixty-four percent of the respondents had experience treating patients with Fabry disease (Table 1). The numbers of patients with Fabry disease treated by respondents were 1 (16.7%), 2–5 (8.8%), 5–10 (2.9%), and ≥10 (1.5%), in that order. The patients with Fabry disease had variant phenotypes (34.0%), followed by classic (30.2%), both (9.4%), and unknown (26.4%) phenotypes (Fig. 1A). In all, 37.1% of nephrologists and 37.5% of cardiologists saw more variant patients than did pediatricians (0%) (Fig. 1B).
Kidney biopsy timing
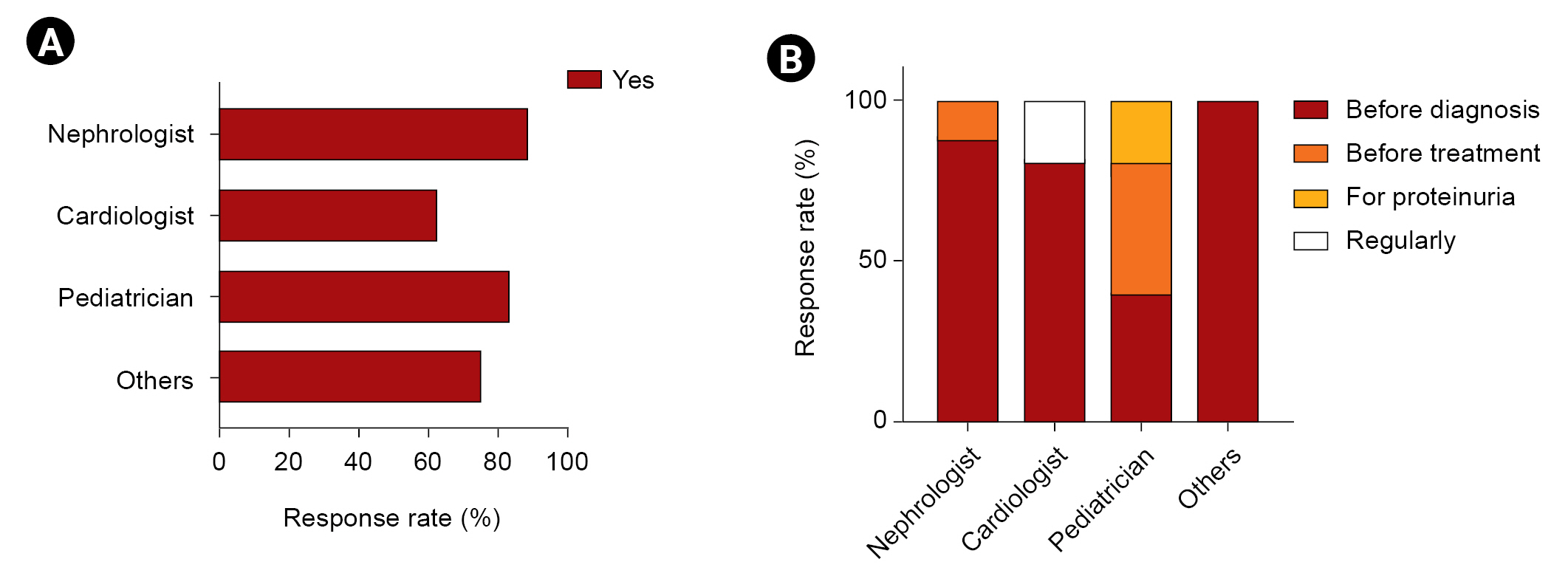
The majority of respondents (46 of 70) indicated that a kidney biopsy should be performed when a diagnosis is made, while 12.9% replied that a biopsy was unnecessary (Table 2). Only 40.7% of non-nephrologists agreed that a kidney biopsy was required at the time of diagnosis, compared with 81.4% of nephrologists. Moreover, 22.2% of non-nephrologists considered a kidney biopsy unnecessary. Cardiologists (62.5%) were less likely than nephrologists (88.5%) and pediatricians (83.3%) to recommend a kidney biopsy (Fig. 2A). The majority of nephrologists and cardiologists would order a kidney biopsy prior to diagnosis, whereas pediatricians responded that a kidney biopsy should be performed prior to both diagnosis and treatment (Fig. 2B).
Need for a Fabry registry
The majority of respondents (79.3% of other experts and 87.8% of nephrologists) said they would take part if KSN created a Fabry registry (Table 2). Two pediatricians and one cardiologist replied that they had no intention of signing up to a Fabry registry.
Assessment of lyso-Gb3
If lyso-Gb3 was covered by Korean health insurance, the majority of nephrologists (86.0%) would measure it after ERT, with smaller proportions choosing diagnosis, screening, and before ERT (Table 2). A majority of non-nephrologists did not agree that lyso-Gb3 should be measured at the time of diagnosis and for screening. Additionally, 60% of nephrologists expressed a preference to monitor lyso-Gb3 every 6 months. If lyso-Gb3 measurement was reimbursed by insurance in nephrology and cardiology, the likelihood that it would be used to determine the response to treatment increased compared with the current state, in which it is not covered (Fig. 3C).

Comparative analysis of specialist agreement on lyso-Gb3 measurement in Fabry disease.
The comparison of agreement of measurement of lyso-Gb3 (A), timing according to responders’ specialty (B). (C) If lyso-Gb3 measurement is insured, diagnosis, before and after treatment lyso-Gb3 measurements were compared to the respondent’s area of expertise. Lyso-Gb3, globotriaosphingosine.
Interval and tests for kidney function in Fabry disease
Half of the responders, particularly nephrologists, selected 3 months as the appropriate follow-up interval (Table 2). In 80.0% and 75.7% of cases, proteinuria and estimated GFR (eGFR) by serum creatinine, respectively, were selected as the optimal kidney function tests. Cardiology and pediatric experts were associated with comparable rates at 3-month and 6-month intervals, respectively, and the follow-up interval for kidney function tests was greater than that indicated by nephrologists (Fig. 4A). Most nephrologists stated that proteinuria, eGFR, α-Gal A activity, cystatin C, and lyso-Gb3 levels should be measured, and kidney biopsies should be conducted to monitor kidney function, although members of other departments infrequently agreed (Fig. 4B).
Definition and tests of Fabry nephropathy
No consensus was seen in the survey results regarding a definition of Fabry nephropathy. When Fabry nephropathy was suspected, physicians measured α-Gal A activity (72.8%). This was followed by kidney biopsy, genetic testing, and measurements of lyso-Gb3 (Table 2).
Treatment of Fabry nephropathy
For additional treatment of ERT in Fabry nephropathy, an angiotensin-converting enzyme inhibitor and/or angiotensin receptor blocker was considered by 82.8% of respondents. A low-protein diet and low sodium intake were considered by 55.7% and 24.3%, respectively (Table 2).
Among responders, the stages of microalbuminuria and proteinuria were identified as the starting point of ERT treatment in patients with variant Fabry nephropathy by 51.7% and 28.5%, respectively. A minority of nephrologists indicated that treatment should be performed if proteinuria was present or eGFR was less than 90 mL/min/1.73 m2. Pediatricians frequently indicated that treatment should be administered when microalbuminuria or proteinuria was once present (Fig. 5). Conversely, nephrologists and cardiologists revealed that the majority of responses were treated during albuminuria, but some indicated that early treatment is required even in the absence of albuminuria.
Discussion
In 1984, ophthalmologists reported the first case of Fabry disease in Korea. In 2019 and 2020, the number of newly registered patients according to the Korean Standard Classification of Disease (KCD) was 28 (8 male and 20 female) and 40 (16 male and 24 female), respectively [17]. Despite the fact that the Korean medical insurance and Korea Disease Control and Prevention Agency had a system for controlling rare diseases, Fabry disease patients were underdiagnosed and untreated for an extended period [16,19,20]. Although Fabry disease has been diagnosed at an increasing rate in recent years, it is still extremely rare and difficult to access by experts [21]. In a French survey of 152 nephrologists, few doctors (22%) directly managed patients with Fabry disease and 18% had made a diagnosis on their own [21].
This study examines the current level of awareness among Korean specialists regarding available treatment options for Fabry nephropathy. The majority of respondents were nephrologists, while pediatricians and cardiologists followed in second and third place, respectively. Nephrologists in Argentina diagnosed half of Fabry disease patients [22]. While nephrologists can also treat patients with classic diseases, they are more likely to treat variants of the disease [23]. Different opinions were held by nephrologists and non-nephrologists regarding the diagnosis and treatment of Fabry nephropathy. Because it is a progressive multisystem disease, the involvement of other vital organs (the heart and brain, in particular) must be determined by an appropriate specialist [24]. In addition, the clinical heterogeneity of Fabry disease necessitates an individualized approach to treatment that is based on the genotype, sex, family history, phenotype, and specific clinical symptom severity of each patient [3,6]. Confirming the opinions of experts about appropriate approaches to the screening, diagnosis, and treatment of Fabry nephropathy is therefore a useful activity.
The majority of nephrologists considered the time of diagnosis optimal for a kidney biopsy, whereas 22.2% of non-nephrologists indicated that a kidney biopsy was unnecessary. Although there was no consensus on a definition of Fabry nephropathy, most respondents chose α-Gal A activity, kidney biopsy, genetic studies, and lyso-Gb3 levels as diagnostic tools. A total of 24% of nephrologists did not agree with the need for a Fabry nephropathy registry. Most experts agreed with measuring lyso-Gb3 as a biomarker. Currently, measurements of lyso-Gb3 are not covered by insurance, so it is being requested by a specific company. If lyso-Gb3 is covered by Korean health insurance, most specialists reported that they would measure lyso-Gb3 after ERT treatment. However, most non-nephrologists agreed that measuring lyso-Gb3 after treatment would be appropriate but would perform such measurements at the time of diagnosis. New biomarkers such as lyso-Gb3 were impaired in patients with normal albuminuria levels [12,17,25] and were more closely correlated with the expression of Fabry nephropathy [18]. Unpublished data from a Korean study by Cho et al. found that lyso-Gb3 is a screening marker that can identify patients who are eligible for a Fabry gene analysis in patients with chronic kidney disease with unknown etiology. This suggests that coverage by insurance should be considered.
Half of the respondents (54.4%), particularly nephrologists, selected proteinuria and eGFR by serum creatinine to measure kidney function every 3 months. Although proteinuria and GFR are considered important markers of Fabry nephropathy, most kidney involvement occurs in a non-proteinuria state [2,3,26]. These findings suggest that the field of expertise of the treating physicians should be given greater attention. Nephrologists evaluate kidney involvement using a kidney biopsy earlier than do non-nephrologists [24,26]. In addition, they treat and monitor kidney involvement more proactively than do non-nephrologists. Consequently, for the treatment of Fabry disease, a discussion with a specialist in the relevant field will be of great benefit.
This is the first survey of Korean experts on Fabry disease and how they manage Fabry nephropathy. Unlike previous Korean patients with Fabry disease studied by a nationwide survey, most patients being treated by survey respondents had a variant form of the disease [16]. The specialty of the diagnosing physician influenced the phenotype of the disease in each patient. Nephrologists and cardiologists treat more patients with variant types, while pediatricians see those with the classic type (Fig. 1). Recent screening studies of Fabry disease in high-risk clinics (1995–2017) confirmed this pattern [23]. Two Korean patients with Fabry nephropathy reportedly progressed to kidney failure with renal replacement therapy in 1989. Recently, a cardiac variant was reported in Korea [27]. Because Korean experience with Fabry disease has been sporadic and intermittent until recently, clinicians are interested in sharing their experiences with the disease [28]. A recent unpooled systematic review found that different patient populations could require different disease-management and therapeutic goals depending on age, genotype, and disease severity and/or level of organ involvement [13]. Although National Health Insurance service benefits set by the KCD (during the 3 months after registration) was 38,903,000/person, the burden of cost remains a barrier for ERT in Fabry disease, particularly for female patients with classic Fabry disease or other patients with the variant type. Fewer reports of ERT in adult females were available compared with those in adult males due to the limitations of retrospective observational and case-series studies [13]. Clinicians, and nephrologists in particular, prefer to monitor kidney manifestations to determine the timing of ERT after diagnosis. Most respondents chose microalbuminuria as the appropriate stage to initiate ERT. An angiotensin-converting enzyme inhibitor and/or angiotensin receptor blocker was considered a non-ERT treatment, followed by a low-protein diet and low sodium intake.
Diagnostic and treatment guidelines for Fabry disease emphasize a multidisciplinary approach to disease management due to multisystem involvement [14,29]. Our findings show that specialists in the field of nephrology have limited expertise with tests and medicines. In fact, a survey of nephrologists in France revealed that knowledge of kidney injury was poor (less than 50% chose the correct answers in a test) [21]. In addition, nephrologists may lack the experience of other specialties when it comes to phenotype or genetic testing; therefore, patients with Fabry disease should receive care from a multidisciplinary team comprising experienced professionals in neurology, cardiology, pediatrics, and genetics, in addition to nephrology.
This research had numerous limitations. First, selection and recall bias is possible in a voluntary survey questionnaire. Second, due to the limited number of specialists with experience in treating this disease, only a small number of respondents completed the survey. Third, responders may have been confused about phenotypic categories because classic and variant phenotypes were not defined in this survey. Fourth, pediatric nephrologists may be considered non-nephrologists due to the nature of their sub-specialty training. Finally, clinicians may diagnose or treat Fabry nephropathy based on the Korean medical insurance indications.
The majority of respondents to the survey were medical professionals who have treated or are currently treating a patient with Fabry disease. They agreed that it is important to initiate ERT early in Fabry nephropathy, particularly in patients with the variant type of the disease, and that lyso-Gb3 is a valuable biomarker, highlighting the importance of insurance coverage for lyso-Gb3 testing.
Notes
Conflicts of interest
All authors have no conflicts of interest to declare.
Funding
This research was supported by the Soonchunhyang University Research Fund.
Data sharing statement
The data presented in this study are available on request from the corresponding author.
Authors’ contributions
Conceptualization, Data curation: SJC, SHK
Formal analysis: SJC, SHK, KHJ
Methodology: SJC, SHK, MSL, EC, EYL
Investigation: ESK, YJK
Software: MSL
Supervision: YJK
Validation: BHC, EHB, EYL
Funding acquisition: SJC
Writing–original draft: SJC, SHK
Writing–review & editing: MSL, SP, EC, SSH, ESK, BHC, KHJ, EHB, EYL
All authors read and approved the final manuscript.
Supplementary Materials
Supplementary data are available at Kidney Research and Clinical Practice online (https://doi.org/10.23876/j.krcp.22.235).