Introduction
Hypertensive nephrosclerosis is usually associated with chronic hypertension, which increases the risk of progressive renal disease. Among patients with malignant hypertension, thrombotic microangiopathy is complicated and associated with renal dysfunction at the time of diagnosis [1], [2]. Thrombotic microangiopathy is characterized by fragmentation of red blood cells (schistocytes) and thrombocytopenia in peripheral blood smears, and serologically, elevated serum lactate dehydrogenase (LDH). Therefore, thrombotic microangiopathy associated with malignant hypertension is important for decision making among hypertensive patients who present with renal failure with thrombocytopenia. We report a case of thrombotic microangiopathy in a young male patient who neglected blood pressure control. This case emphasizes the importance of early recognition of renal failure and thrombocytopenia among patients with uncontrolled hypertension.
Case report
An 18-year-old Asian man was admitted to the emergency unit complaining of general weakness, generalized edema, and dyspnea over the past 3 days. He was diagnosed with hypertension of uncertain degree at a school health screening 1 year ago. However, he had not sought medical attention and had never taken antihypertensive medication. His vital signs were normal except for uncontrolled blood pressure up to 190/120Ā mmHg. Physical examination revealed coarse breath sounds with moist rales. Laboratory findings revealed a white blood cell count of 10,870/mm3, hemoglobin of 10.9Ā g/dL, platelet count of 75,000/mm3, reticulocyte count of 8%, blood urea nitrogen level of 82Ā mg/dL, serum creatinine level of 7.66Ā mg/dL, and LDH of 3,634 IU/L. Urinalysis showed 4+ proteinuria and microscopic hematuria. Urinary protein/creatinine ratio was 5,239Ā mg/g. Serological tests revealed that he was negative for hepatitis B virus antigen and hepatitis C virus antibody, complement component C3 103.1Ā mg/dL, complement component C4 19.2Ā mg/dL, and negative for antinuclear antibody, P- and C-antineutrophil cytoplasmic antibody. Renal Doppler ultrasonography revealed that renal perfusion was decreased and echogenicity was diffusely increased with swelling. The right and left kidneys were 10.5Ā cm and 10.2Ā cm in size, respectively (Fig. 1). Next, we performed a peripheral blood smear because of thrombocytopenia and increased LDH levels. Blood smear showed an increased number of schistocytes (15ā20/high-power fields). We diagnosed acute kidney injury due to thrombotic microangiopathy.
We decided to start high-dose steroid therapy and plasmapheresis until available results of a disintegrin and metalloproteinase with a thrombospondin type I motif, member 13 (ADAMTS13) activity. Therefore, we performed plasmapheresis 11 times. Follow-up blood smear showed a decreased number of schistocytes and recovered platelet count and serum LDH level. During plasmapheresis, blood pressure was uncontrolled up to 200/110Ā mmHg. Therefore, we prescribed antihypertensive medication including irbesartan, carvedilol, and nifedipine. His blood pressure was stabilized to 140/90Ā mmHg after intensive blood pressure control. Two weeks after admission, ADAMTS13 activity was reported in the normal range (66%, normal 44ā121%). We discontinued plasmapheresis and gradually tapered the prednisolone dose.
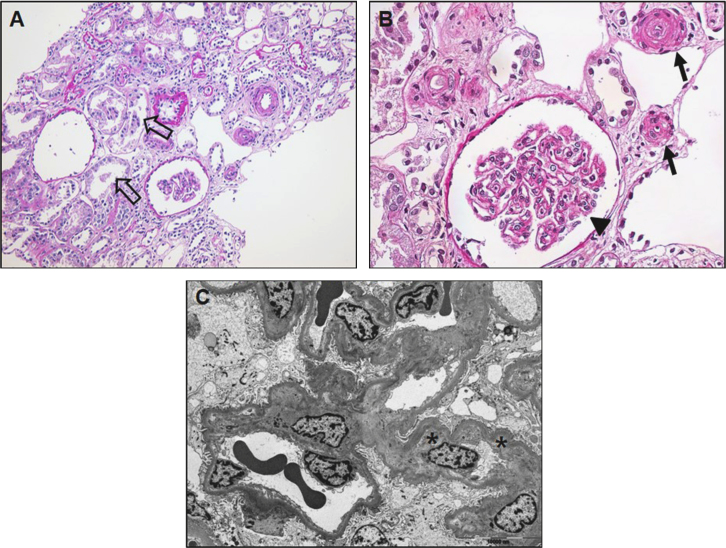
We evaluated the secondary cause of hypertension. On ophthalmoscopic examination, we found early-stage hypertensive retinopathy with hard exudate. On abdominal magnetic resonance imaging, there was no evidence of renal artery stenosis or adrenal mass such as pheochromocytoma or adrenal cortical adenoma. We performed renal biopsy for evaluation of acute kidney injury and proteinuria. Renal biopsy revealed mild denuded tubular cells, ischemic glomerular changes with wrinkling of glomerular capillary walls, and hyperplastic arteriolitis with onion skin appearance. These pathological findings were consistent with hypertensive nephrosclerosis associated with thrombotic microangiopathy (Fig. 2). At 10 months of initial presentation, serum creatinine level was 2.43Ā mg/dL and urinary protein/creatinine ratio was decreased to 100Ā mg/g after intensive blood pressure control. Until now, blood pressure was stable at approximately 130/80Ā mmHg with taking antihypertensive medication.
Discussion
We present a case of acute kidney injury due to thrombotic microangiopathy associated with malignant hypertension. Our patient showed clinically severe hypertension with mild hypertensive retinopathy and thrombocytopenia. Laboratory and pathological findings were consistent with thrombotic microangiopathy with hypertensive nephrosclerosis. These conditions may have resulted from uncontrolled blood pressure.
Despite the relatively low incidence of renal disease, chronic hypertension is known as a risk factor for development of renal disease. Lindeman et al [3] have reported that there is an inverse relationship between blood pressure and decline of renal function in the range of high blood pressure. The followings have an increased risk of development of hypertensive nephrosclerosis: blacks, aging, smokers, and patients with uncontrolled hypertension, atherosclerosis, and underlying chronic kidney disease such as diabetic nephropathy [4]. Interestingly, our patient had a relatively short duration of hypertension at a young age and he never received medical treatment for uncontrolled blood pressure. These conditions may have led to hypertensive nephrosclerosis.
Pathological changes in hypertensive nephrosclerosis are characterized by vascular, glomerular, and tubulointerstitial lesions [5]. Vascular lesions consist of hyalinosis in benign nephrosclerosis and muscular hyperplasia in the arcuate and interlobular arteries and afferent arterioles in malignant nephrosclerosis, which lead to vascular luminal narrowing [5], [6]. The glomerular lesions in hypertensive nephrosclerosis are characterized by three categories: normal in morphology; ischemic glomeruli with capillary collapse, and retraction of the glomerular tuft; and hypertrophic glomeruli with segmental scar or hyalinosis [6]. Tubulointerstitial damage is associated with chronic hypoxia resulting in ischemic damage in the tubulointerstitium [6]. In our case, we could not find arteriolar thrombosis and fibrinoid necrosis. However, electron microscopic findings showed subendothelial electron lucent expansion and increased electron lucency in the mesangial and paramesangial areas, suggestive of endothelial cell injury due to accelerated hypertension. In light microscopy, glomerular lesions showed a varying degree of wrinkling of the capillary walls, which suggest ischemic changes in the glomeruli. Therefore, these biopsy findings represent thrombotic microangiopathy associated with hypertensive nephrosclerosis.
In conclusion, this case emphasizes the importance of early recognition of renal failure and thrombocytopenia among patients with uncontrolled hypertension. Early intervention such as plasmapheresis and intensive blood pressure control might be helpful in the prevention of deterioration of renal function.




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print
















